


The most frustrating thing about clay trails is how dramatically a little (unfrozen) water can transform them. Dry or frozen they’re smooth, firm and high-traction- a joy to ride.

I held it together and rode through, clear down to the trailhead and my car. Wasting no time, I heaved the mud-caked bike (weighing probably 40 pounds) atop my vehicle, and drove straight to the do-it-yourself carwash. Foothill-clay-mud is almost impossible to wash off with an ordinary garden hose; I needed the power and warm water of the carwash blaster-nozzle.
Tangent: Yes, yes I know: taking your mtn bike to the carwash is a huge no-no. Water gets in seals, messes up bearings, yada, yada. But like many mtn bikers, I am an occasional closeted carwash visitor. It’s just so much easier and time-efficient than any other conceivable method to remove clay from your bike. I always feel sort of embarrassed, guilty and yet resigned all at the same time when I do it, sort of like a high-school kid who’s just completed an abstinence-only sex ed class: I know the safest thing is not to do it, but simultaneously am aware that given the combination of need and opportunity, I almost certainly will.

Tangent: Utah mud is a surprise to transplants like me. In other parts of the country, mud is no big deal. People ride through it all the time. In fact mountain bike magazine reviews of tires routinely include descriptions of “mud-shedding” capability. In Utah there’s no such thing. No bike tire sheds mud. A “mud-shedding tire” is an oxymoron.
Utah clay-mud is an even bigger concern for backcountry drivers. Countless unpaved roads in Southern and Western Utah are completely impassable when wet. When I drive deep solo into the backcountry, I generally pack along a mountain bike as an escape pod, meaning that if I break down or get stuck, I can reasonably bike 50, 60, 80 miles or more to pavement and assistance. But in mud, my escape pod is as immobile as my vehicle; there’s nothing to do but wait, either for sun (to dry) or cold (to freeze.)
Ranchers and other folks who routinely drive around the Utah backcountry in fall/spring/winter typically bring along tire-chains, and if you look closely at deep, dried-out tire-ruts on many desert-clay roads, you’ll noticed that they’re cross-hatched with chain-marks.
All About Clay

Mineral particles are characterized as sand, silt or clay, and all are basically teensy-weensy chips of rock. Rocks are constantly being eroded and broken down, and these smaller particles are products of this process.
Extra Detail: Factors in rock erosion include of course wind, sun and water. You may think you already “get” the erosive effect of water on rocks: waves, streams and raindrops repeatedly pound on exposed rock, and when frozen, water-ice expands in volume, expanding and propagating cracks and crevices. But water- specifically rain- also has a corrosive chemical effect on rock. Raindrops (H2O) combine with Carbon dioxide(CO2) in the atmosphere to create Carbonic acid (H2C03). Over time, the positively charged hydrogen atoms in the acid molecules replace other positively-charged elements in rocks like limestone and granite, causing them to break into smaller pieces.
Lichens also play an important role in the creation of mineral particles and ultimately soil. ![Lichen Genera Antelope[4] Lichen Genera Antelope[4]](https://blogger.googleusercontent.com/img/b/R29vZ2xl/AVvXsEhcyeqB4DIiA1uWZPg83I4lR1pxcJ5O8BO2m3M17lFoIP-0gbzPyQczvT8g3WiMaKmuKmxFWHSBiJckZC926ayhfYyQ4VFiX4OTQxarMgDGlU4eyBIfQT6YlrF3Z8Gb0TuK9RcUntLZScY/?imgmax=800)
The primary difference between sand, silt and clay particles is size. A grain of sand ranges from 0.05mm to 2mm in diameter, while a grain of silt is anything between 0.05mm and 0.002mm. Clay particles are those under 0.002 mm in diameter.
Note: No, I don’t have an electron microscope. The electron microscopy shots in this post were pulled from this extremely informative slide deck on the Washington State University Dept. of Crop and Soil Sciences website.

*My calculation, could be wrong.
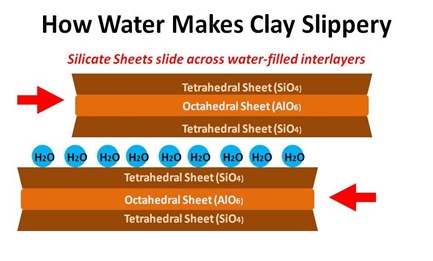
Up close, clay particles are generally flattish flakes, which means that have more surface area for their size. They’re typically negatively charged and the combination of negative charge and large surface area means that they bond easily to water molecules. Saturated sand is about 20% water. Saturated clay is about 48% water.
When you walk out of the water onto a sandy beach, wet sand sticks to your bare feet. But if you lie on the dry sand for a few minutes, the sun and wind will dry the water molecules holing the sand grains together and to your skin, and the sand will easily brush off. If you’re impatient, you can walk up to one of those beachside showers or hoses and quickly rinse off. The sprayed water drops will easily penetrate the large gaps between the roundish sand-grains, pushing them apart and away.
But if you walk barefoot in wet clay, and then lay back in the sun, your feet will take much longer to dry. The much smaller spaces between flakes are more difficult for sun and wind to penetrate. And when, eventually, the clay does dry, it’ll still take more effort to dislodge than dry sand; the tiny flattish flakes make almost a little concrete later on your skin. More frustratingly, if you walk over to a shower or hose, it’s way, way harder to wash the clay off. The sprayed water drops have great difficulty penetrating the much smaller gaps between clay-flakes, which are already filled/clogged with adhering water molecules.

The prevalence of clay in foothill soils is not only a factor for mountain-bikers, gardeners and builders, but also for wild plants. Clay soils (particularly saturated clays soils) take longer to warm up in the spring, and it’s likely that the heavy clay content in the soil of the Wasatch foothills delays spring blooms by a week or two.
Well, that all makes sense. The small flat particles of clay make it sticky, hard to wash off and generally problematic for mtn bikers. But if it’s so sticky, why is it so slippery? To understand this one, we have to know something about the composition and structure of clay.
The Structure of Clay
Clay is a sheet silicate, meaning a silicon-based substance that forms into 2-dimensional crystalline sheets. Silicon is a common element in rocks, and hence clay. Silicon, atomic #14, has 4 open spots in its 3rd electron shell, and so “wants” to make 4 electron bonds. Another abundant element is of course Oxygen, atomic #8, which has 2 open spots in its 2nd electron shell. Together, Silicon and Oxygen comprise Silicon Tetraoxide, SiO4, in which the single Silicon atom binds to each of 4 Oxygen atoms, which each have 1 bond with the Silicon atom, and another bond with a fellow Oxygen atom. Together the 5 atoms form a tetrahedron*, with an Oxygen atom at each corner and the Silicon atom in the center.
*A tetrahedron is 4-sided, with a triangle on each face. It looks, at first glance, sort of like a pyramid, but it’s not. A pyramid is 5-sided, with triangles on 4 sides, and a square on the 5th/base.
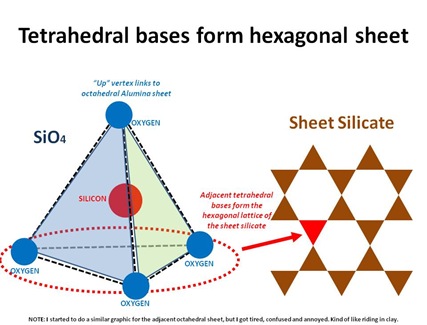
OK, now the pointy-“up” corner of the tetrahedron is connected to an Aluminum molecule, together with other tetra-vertices and bonded hydroxyls (OH) to form AlO6, an octahedron (8-sided). So the tetrahedral Silica layer lies adjacent to an octahedral Alumina layer. At a molecular level then, clay is basically a bunch of stacked, alternating sheets, which is why the flakes are flattish under the electron microscope.
That’s the basics. From here things start to get complicated, and diverge into different clay types. The biggest difference is between so-called 1:1 clays, which alternate tetrahedral and octahedral layers, and 2:1 clays, which consist of octahedral layers sandwiched between 2 tetrahedral layers. The other big differentiator is something called isomorphic substitution, which is a fancy term for the replacement of a Silicon or Aluminum atom by something else, such Aluminum (in the tetrahedral sheet) or Magnesium (in the octahedral layer).



4 comments:
March will break your heart.
Super post. I never cuss more than when I get trapped in that stuff because I forgot it was there or assumed it had dried out.
Nice road bike!
150mm stem, V brakes, road tires, 22 inch bars. Must be fast!!
Sorry man, couldn't help but give you some grief. Some of us are roadies who mountain bike, and some of us are just (slow) dudes who ride off road.
Road bike?! That's it- I'm posting some road-riding helmet-cam footage...
Actually, the front tire is a 2.4". I think my monster Wal-Mart gloves make it look narrow. Stem is 120mm,which helps with the 1,500 - 2,000 ft climbs typical on summertime rides around here. And yes I have long ago fessed up to my V-fetish.
Bars = 24". I tried 26" on another bike a few years back and switched back. Once the higher altitude singletrack opens up in the Wasatch, the wider bars are tough to fit between the aspens at speed...
All our bikes are evolved to fit rider, terrain and riding style. Wait a minute..is it evolved, or intelligently designed;)
Great post today, I've learned so much here my riding buddies will be cursing my ability to talk and ride at the same time!
Post a Comment